Ethylene Sulfate: Uses, Properties and Benefits
Ethylene sulfate is an important organic chemical compound widely used in industrial and electrochemical applications. It is mainly known for its role in lithium-ion battery technology, where it works as an electrolyte additive to improve battery efficiency and stability. With the growing demand for rechargeable batteries in electric vehicles, smartphones, and renewable energy systems, the importance of ethylene sulfate has increased significantly. This compound helps improve battery life and enhances overall performance, making it a valuable material in modern industries.
What is Ethylene Sulfate?
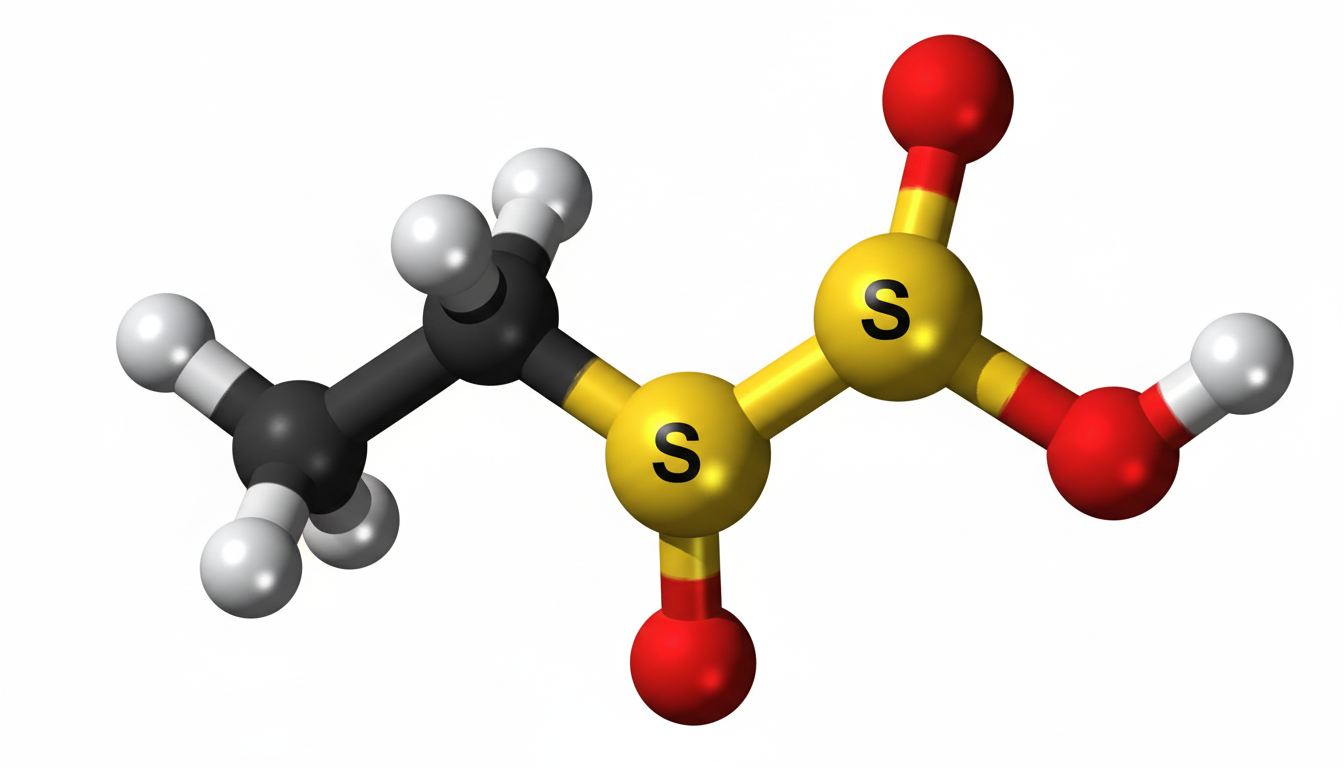
Ethylene sulfate is a cyclic sulfate compound that contains sulfur, oxygen, carbon, and hydrogen atoms in its chemical structure. It is mainly used in advanced electrochemical systems because of its unique chemical behavior. The compound is specially designed to improve the performance of lithium-ion batteries by forming a protective layer on the electrode surface. Due to its high effectiveness, it has become an important material in energy storage research and industrial battery manufacturing.
Chemical Structure of Ethylene Sulfate
The chemical structure of ethylene sulfate consists of a cyclic ring formed by sulfur and oxygen atoms attached to an ethylene group. This structure gives the compound its reactive and stable properties required for electrochemical applications. The cyclic arrangement allows the compound to interact efficiently with battery electrodes during charging and discharging processes. Its molecular design plays a major role in improving the formation of stable electrolyte layers inside lithium-ion batteries.
Physical Properties of Ethylene Sulfate
Ethylene sulfate has several important physical properties that make it suitable for industrial applications. It usually appears as a colorless or light-colored liquid depending on purity and storage conditions. The compound has good solubility in organic solvents and remains stable under controlled conditions. It is known for its high chemical compatibility with lithium salts and battery electrolyte materials. These physical characteristics help improve battery performance and increase operational efficiency.
Chemical Properties of Ethylene Sulfate
The chemical properties of ethylene sulfate make it highly useful in electrochemical systems. It is reactive under charging conditions and helps create a stable protective layer on battery electrodes. This property prevents harmful side reactions and improves battery durability. Ethylene sulfate also has good oxidation resistance, which supports stable performance during long battery cycles. Its controlled reactivity makes it an effective additive in modern energy storage technologies.
Working Principle of Ethylene Sulfate
Ethylene sulfate works by decomposing on the electrode surface during the initial charging cycles of a lithium-ion battery. This decomposition forms a thin protective film known as the solid electrolyte interphase (SEI). The SEI layer protects the battery electrode from continuous chemical reactions that could reduce battery life. As a result, batteries using ethylene sulfate show better charge retention, improved safety, and enhanced overall performance compared to systems without this additive.
Applications of Ethylene Sulfate
Ethylene sulfate is widely used in lithium-ion battery manufacturing for portable electronics, electric vehicles, and renewable energy systems. It is also used in electrochemical research laboratories to study battery behavior and improve energy storage technology. In industrial chemistry, the compound is sometimes used in specialized synthesis processes where controlled chemical reactions are required. Its growing use in advanced battery systems has made it an important material in modern technological industries.
Importance in Lithium-Ion Batteries
The role of ethylene sulfate in lithium-ion batteries is extremely important because it improves battery stability and efficiency. It helps form a strong and stable SEI layer that reduces electrolyte decomposition and prevents capacity loss. This increases battery life and allows the battery to operate safely over long periods. Due to these benefits, many modern battery manufacturers use ethylene sulfate to enhance battery quality and performance.
Advantages of Ethylene Sulfate
Ethylene sulfate provides several advantages in battery technology and industrial applications. One major benefit is improved battery cycle life, which allows rechargeable batteries to last longer. It also enhances battery safety by reducing unwanted chemical reactions inside the cell. Another advantage is better thermal stability, which helps batteries operate effectively under different temperature conditions. Its ability to improve energy efficiency makes it highly valuable in modern electrochemical systems.
Limitations of Ethylene Sulfate
Despite its many advantages, ethylene sulfate also has some limitations. It is a reactive chemical compound that requires careful handling and controlled storage conditions. Improper usage can sometimes lead to unwanted reactions that may affect battery performance. Additionally, the production process of ethylene sulfate can be expensive because it requires specialized industrial methods. These factors can increase manufacturing costs in some applications.
Safety and Handling of Ethylene Sulfate
Proper safety measures are important when handling ethylene sulfate in industrial or laboratory environments. The compound should be stored in sealed containers away from moisture, heat, and direct sunlight. Protective equipment such as gloves, masks, and safety goggles should be used during handling to avoid direct exposure. Since it is a reactive material, only trained professionals should use it in controlled conditions to ensure safety and prevent accidents.
Environmental Impact of Ethylene Sulfate
The environmental impact of ethylene sulfate depends on how it is handled and disposed of after use. If released improperly, chemical compounds used in industrial processes may affect soil and water quality. Therefore, industries must follow proper disposal and recycling methods to minimize environmental risks. Research is also being conducted to develop safer and more environmentally friendly battery materials for future applications.
Future Potential of Ethylene Sulfate
The future potential of ethylene sulfate is very promising because of the increasing demand for advanced battery technologies. As electric vehicles and renewable energy systems continue to grow, the need for efficient and long-lasting batteries will also increase. Researchers are working on improving the performance and cost-effectiveness of ethylene sulfate for next-generation energy storage systems. Its role in developing safer and more powerful batteries makes it an important compound for future technological advancements.
Conclusion
Ethylene sulfate is a highly important chemical compound used mainly in lithium-ion batteries and electrochemical industries. Its unique chemical and physical properties help improve battery efficiency, stability, and safety. Although it has some limitations, its advantages in energy storage technology are significant. With ongoing research and increasing demand for rechargeable batteries, ethylene sulfate is expected to remain a key material in modern industrial and technological applications.

